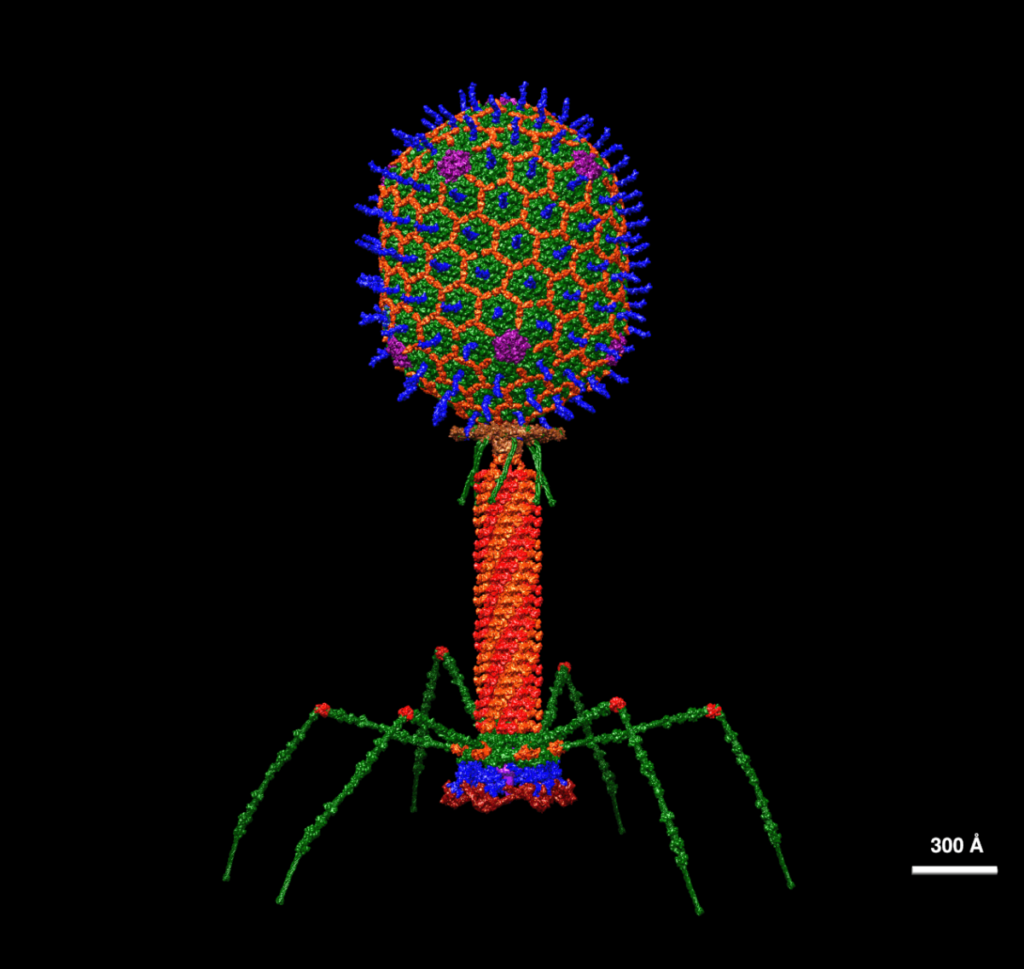
噬菌體療法(Phage Therapy),在細菌紛紛對抗生素產生抗藥性的時候,就成為這些幾乎完全絕望的患者,最後的一絲希望。
Phage 是bacteriophage 的簡稱。
在古希臘文中,bacterio(bacteria) 是「細菌的」,而phagein 則是「噬吃」(to eat),合起來就是「噬菌體」,意思是專以細菌為食的病毒(viruses),有非常非常多的種類。
飲水人喝了不只不會有事,而且還有益健康,那是因為水中有很多噬菌體,為我們控制了細菌的量和種類的緣故。
細菌碰到抗生素,能很快突變演化出能抗藥的細菌出來。
在細菌快速突變演化時,噬菌體這種病毒,也會快速突變演化,可以繼續以細菌為食,這就是噬菌體療法比抗生素強的地方,是在病毒這個奈米級世界的生物療法。
生物學家估計,噬菌體在每48小時內,就能殺死了一半的細菌。這個世界如果沒有噬菌體來不斷維持微生物的生態平衡,那是無法想像的。
抗生素會殺死一堆不同的細菌,但是噬菌體則能更精確地殺死某些種細菌,而對其他的無害,這使得噬菌體療法的副作用更低。
噬菌體在1910年代就發現了,但是隨著在1935年發現磺胺類的藥品(sulfonamide),接著在1942年發現了盤尼西林(penicillin),噬菌體的抗菌特性,除了在蘇聯及東歐繼續有些研究,基本上在世界上絕大多數的地方都被冷落了。
In 1923, George Eliava, from the country of Georgia, traveled to the Pasteur Institute in Paris, where he met d’Herelle. In 1923, he founded the Eliava Institute in Tbilsi, Georgia, which today remains the epicenter of phage therapy. From 2012 to 2014, more than five thousand patients visited the institute’s Phage Therapy Center for treatment. These folks had a variety of bacterial infections, including those caused by antibiotic-resistant bacteria, such as methicillin-resistant Staphylococcus aureus (MRSA). They claim that more than 95 percent of these patients showed significant improvement. Today the Phage Therapy Center continues to welcome patients from around the world.
大意是說:
在喬治亞的「喬治·艾萊瓦噬菌體、微生物和病毒學研究所」(George Eliava Institute of Bacteriophage, Microbiology and Virology),也稱為提比里西研究所,Tbilisi Institute,是目前研究噬菌體療法的中心。(~維基百科)
2012到2014年間,超過5000病患求助於該中心,以治療對抗生產生抗藥性的細菌,包括有抗藥性的金黃葡萄球菌:超級細菌Staphylococcus aureus (MRSA)。據稱超過95%以上的病患都獲得了改善。
The first studies of phage therapy in humans that met FDA standards have now been done. So far, the results have been promising. Phages have successfully treated otitis externa caused by Pseudomonas aeruginosa, a chronic bacterial ear infection that is notoriously difficult to cure; diarrhea; and infected ulcers associated with leg veins. Properly controlled clinical trials have also been carried out, or are in the planning stage, for the treatment of burn wounds and diabetic foot infections. 2 In June of 2015, the European Medicines Agency (EMA) hosted a workshop on the therapeutic use of bacteriophages. One month later, the National Institutes of Health hosted a similar workshop. That same year, the NIH’s National Institute of Allergy and Infectious Diseases announced that phage therapy was one of seven prongs in its plans to combat antibiotic resistance.
大意:第一個用在人體,符合FDA嚴格要求的噬菌體療法而治癒的,是針對由綠膿桿菌(Pseudomonas aeruginosa)引發難纒的外耳炎;這種菌也會引發嚴重腹瀉、腿部靜脈發炎等等。
將噬菌療法用於治療燒燙傷的傷口及糖尿病患的足部等感染,也是正在進行中的計劃。
Anecdotal reports continue to surface of what appear to be miraculous cures of patients with life-threatening infections caused by antibiotic-resistant bacteria. One such recent case, published in 2017 in the journal Antimicrobial Agents and Chemotherapy, describes a sixty-eight-year-old diabetic patient with an overwhelming infection caused by Acinetobacter baumannii, which was resistant to all antibiotics. Out of desperation, Dr. Robert Schooley, head of the Infectious Diseases Division at the University of California, San Diego (USD), enlisted the help of bacteriophage experts, who tailored a cocktail of phages that were active against this bacterium. The phages were administered intravenously—a first. And they saved the patient’s life. (You read about A. baumannii in chapter 15. It is one of a growing number of bacteria for which there are no available antibiotics.) This remarkable anecdotal case, coupled with a number of similar cases seen elsewhere, spurred UCSD to launch a clinical center in 2018 to refine phage treatments and help companies bring them to market.
大意:噬菌體的治療不斷有傳奇性的療癒案例。有位68歲的糖尿病患者,被無抗生素可用的鮑氏不動桿菌 (Acinetobacter baumannii)感染。經過靜脈注射了幾種噬菌體的組合液之後,挽回了生命。
Another remarkable case was reported in 2019. A fifteen-year-old girl with cystic fibrosis also had an antibiotic-resistant Mycobacterium abscessus infection, which developed after a double lung transplant. Not only were antibiotics ineffective, but because of the drugs given to her to prevent organ rejection, her immune system was less able to fight the infection. She was given genetically engineered bacteriophages, and she had a full and stunning recovery from the infection.
大意:另外一個例子,是位移植雙肺的15歲女童,產生囊腫纖維症,並被對抗生素有抗藥性的膿腫分枝桿菌所感染,而抗排斥藥物則進一步降低了她免疫系統的功能。經過基因改造的噬菌體治療之後,那位女童神奇地康復了。
……
除了治療疾病,噬菌體用來處理食物中的細菌,也就是殺菌,也是未來一大應用的方向。
噬菌體療法重新受到重視,就讓我想到這個世界的相生相剋。
細菌再厲害,演化抗藥性的速度再快,在這個世界上,還有一種叫做噬菌體的病毒,基因變化的速度也很快,可以用來抑制細菌,讓這個世界在微生物的世界,保持一個生態的動態平衡。
水中就有很多噬菌體,要健康就要適度多喝水,應該也是基於同一種道理。
有些噬菌體進入了細菌內,並不會馬上殺死細菌,而是在細胞質中形成獨立的質體(Plasmid),甚至對細菌提供特定的功能。而往往是細菌本身變弱的時候,這些潛伏在質體的噬菌體才會快速增生,最終撐破而殺死了細菌。
在微觀的世界,生生滅滅也真是奇妙。
*:Philip K. Peterson. M.D., Microbes —the life-changing stories of germs, 2020, Prometheus Books
P.S. 照片來自維基百科,Atomic structural model of bacteriophage T4 in UCSF Chimera software using pdbs of the individual proteins.,作者:Dr. Victor Padilla-Sanchez, PhD
2024/10/21 噬菌體 Damakey
